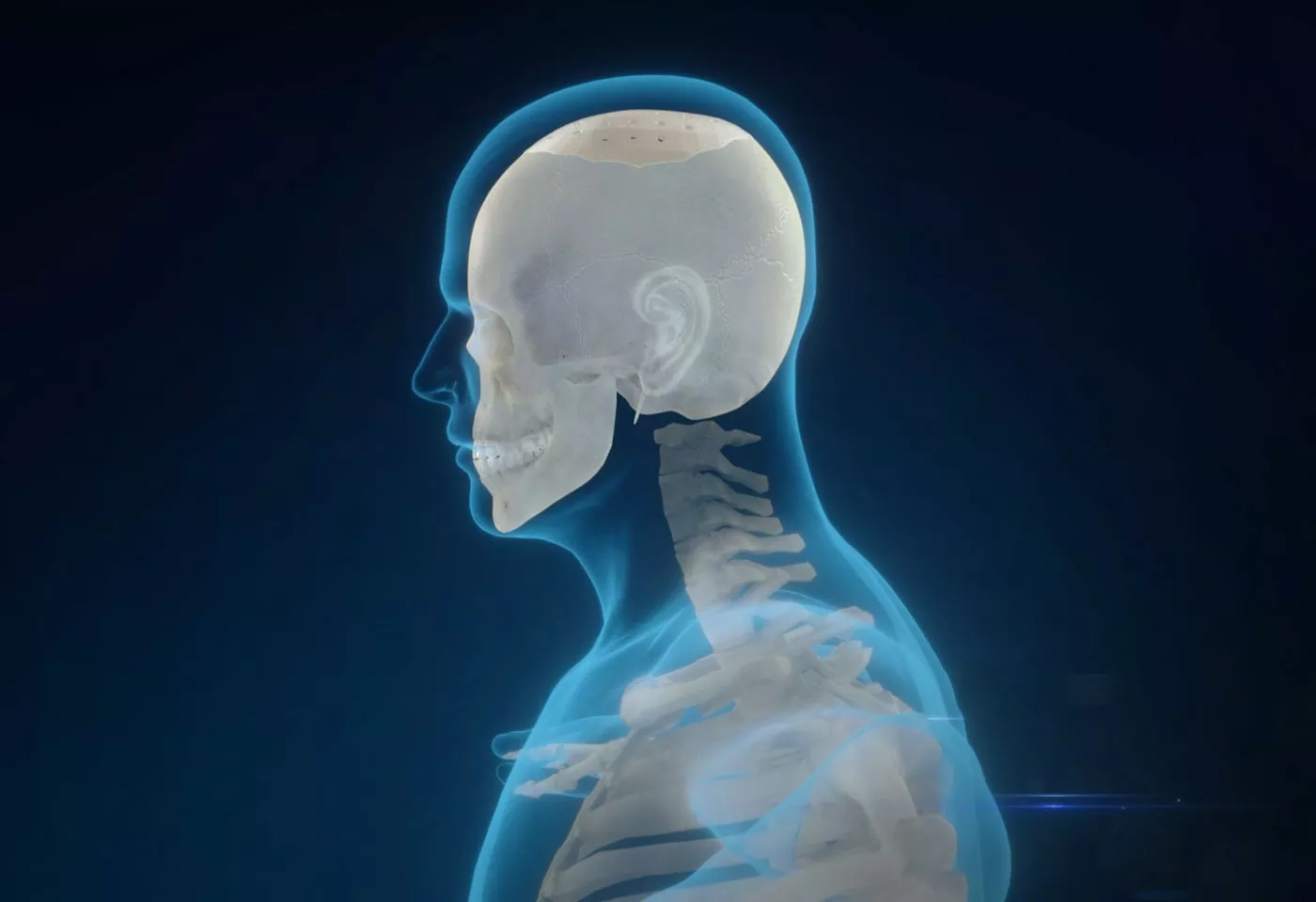
Solution Provider of Medical Biomaterials
Scroll Down
Company Profile
As a subsidiary of Zhejiang Pfluon Technology Co. Ltd., Zhejiang Bio-PAEK Technology Co., Ltd. specializes in the R&D, manufacturing and sales of polymer biomaterials.
The Polymer Division of Pfluon has accumulated plenty of experience in chemical synthesis of PAEK, refining and purification, granulation and modification, injection molding and processing, profile processing, composite materials, application technology as well as other fields. On this basis, Bio-PAEK Technology has expanded the application of PAEK from industrial circle to medical biomaterials industry, thereby achieving the localization of medical biomaterials of PAEK .


Scientific Research Achievements
In 2019, Pfluon cooperated with Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Department of Chemical and Biological Engineering, Zhejiang University, Canwell MEDICAL Co., Ltd., Fudan University, Zhejiang University of Technology and Shanghai Jiao Tong University to jointly undertake the national key R&D projects of the Ministry of Science and Technology, i.e., Research and Development of Key Technologies for Mass Production of Medical PAEK Materials and Surface Biomimetic Modification Technology for Orthopedic Implants.
Therein, Pfluon is in charge of the sub-project Development of New Medical-grade PAEK Materials and Research on the Mass Production of PEEK. Four key technologies have been accomplished in this project, i.e., Precipitation and Granulation Technology in Polymeric Liquid Kettle, Key Technology of PEEK Purification, Preparation of PAEK with High Osteogenic Activity and Preparation of High-strength PAEK. Besides, it has passed nine tests of performance evaluation and biocompatibility evaluation (GB/T16886) in the standard specifications (YY0660-2008) of PEEK polymer for surgical implants. Currently, it is carrying out clinical trials of spinal fusion cage, and the project is scheduled to be accomplished in the first half of 2023.
Performance Strengths
Performance Advantages: PAEK is one of the most comprehensive and suitable biomaterials for human implantation, and it is a gift from heaven to mankind. PAEK enjoys the following nine performance advantages in comparison with metals or other polymers.
Elastic Modulus
Biocompatibility
Mechanical Strength
Wear Resistance
Thermal Conductivity
Fatigue Performance
Medical Imaging Performance
Disinfection property
Easy Molding and Processing Performance
Product Series
After more than 10 years of dedicated research and development, Pengfulong has created the seven major Implong ™ Series of biomaterial products.
Inpulong™(Implong) in Chinese means the popularization of genetic technology and the prosperity of business, while the English trademark Implong implies Implant Long, meaning “long-term implantation”. We aim to contribute to the domestication of biomaterials in China.
Implong CP
Implong™ Coarse Powder Series can provide raw powder for customers with granulation modification or further reprocessing.
Implong FP
Implong™ Fine Powder Series can be used for molding, 3D powder printing, spraying and unidirectional tape prepreg.
Implong GP
Implong™ Pure Granule Series can be used for profile extrusion, plastic monofilament extrusion and injection molding.
Implong CP
Implong™ Compound Granule Series can be used for profile extrusion, plastic monofilament extrusion and injection molding.
Implong SR
Implong™ Stock Shapes - Rod Series can be used for profile-forming machine.
Implong SP
Implong™ Stock Shapes - Plate Series can be used for profile-forming machine.
Implong CFC
Implong™ Continuous Fiber Composite Series can be used for molding, machine work, plastic insert molding, 3D printing, laser winding engineered shaping.
Implong Medical Materials Classification
| Brand Name | Description | Naming Description | Application Process | Application Fields |
| Implong CP Series | Implong™ Coarse Powder Series | CP:Coarse Powder | Blending modification | All the medical equipment that made by granulation modification or further reprocessing |
| Implong FP Series | Implong™ Fine Powder Series | FP: Fine Powder | molding, 3D powder printing, spraying and unidirectional tape prepreg | Ribs, skull, joints, bone plates |
| Implong PG Series | Implong™ Pure Granule Series | PG: Pure Granule | injection molding, extrusion | Fixed nail |
| Implong CG系列 | Implong™ Compound Granule Series | CG: Compound Granule | injection molding, extrusion | Maxillofacial bone |
| Implong SR Series | Implong™ Stock Shapes - Rod Series | SR: Stock Shapes - Rod | machine work | vertebral body fusion cage |
| Implong SP Series | Implong™ Stock Shapes - Plate Series | SP: Stock Shapes - Plate | machine work | vertebral body fusion cage, cranial bones |
| Implong CFC Series | Implong™ Continuous Fiber Composite Series | CFC: Continuous Fiber Composite | molding,laser winding engineered shaping , 3D printing | joints, bone plates |
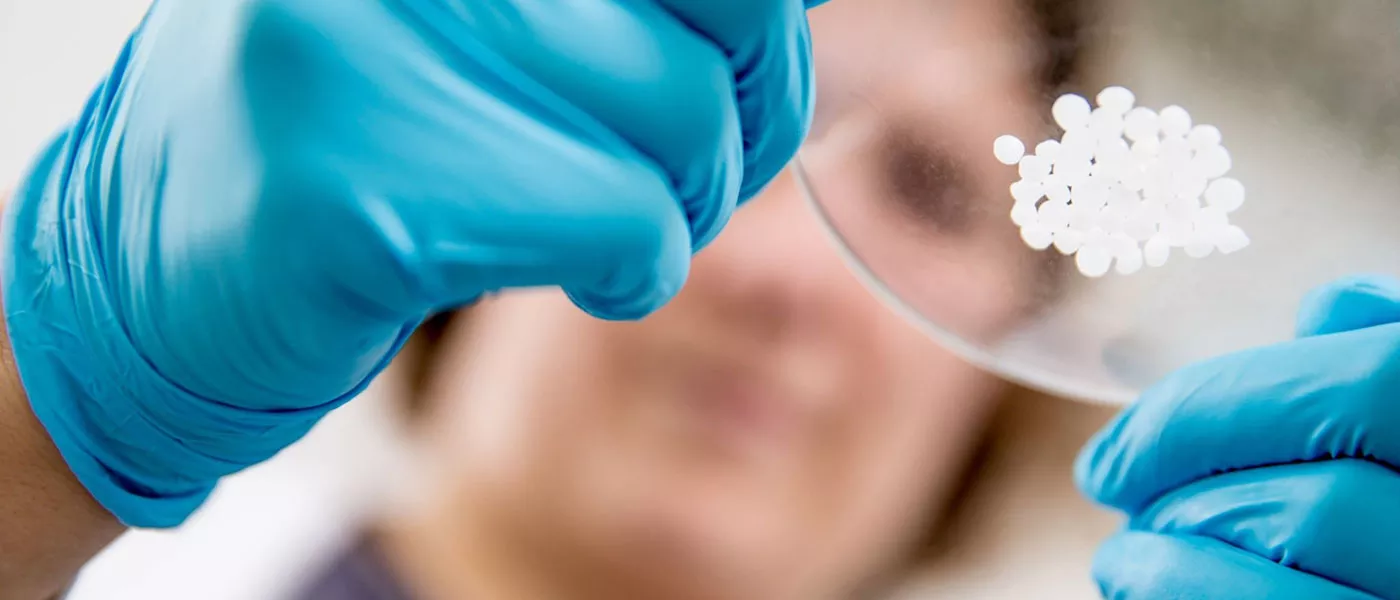
In order to achieve commercial production for the seven Implong ™ Series, Zhejiang Bio-PAEK Technology Co., Ltd. has built PAEK R&D center, medical biomaterials laboratory, 600 square meters of class 10000 clean room, double screw granulator for imported medical materials, single screw extruder for imported medical materials, medical material annealing oven, medical PAEK unidirectional tape prepreg production line, etc. And based on the advanced hardware facilities, we has established ISO13485 quality management system.
Based on the project of “Key Technologies for Mass Production of Medical PAEK and the R&D of Surface Biomimetic Modification Technology for Orthopedic Implants” by the Ministry of Science and Technology, We will continue to conduct comprehensive testing of the performance evaluation and biocompatibility evaluation (GB/T16886) for Standard Specification for Polyether Ether Ketone Polymer for Surgical Implants in Industrialized Biomaterial Products (YY0660-2008). At the same time, we will apply for the document filing in the Medical Device Evaluation Center of the National Drug Administration. And we will cooperate with leading domestic PAEK orthopedic implant device manufacturers to make domestic medical PAEK to enter the market for services.